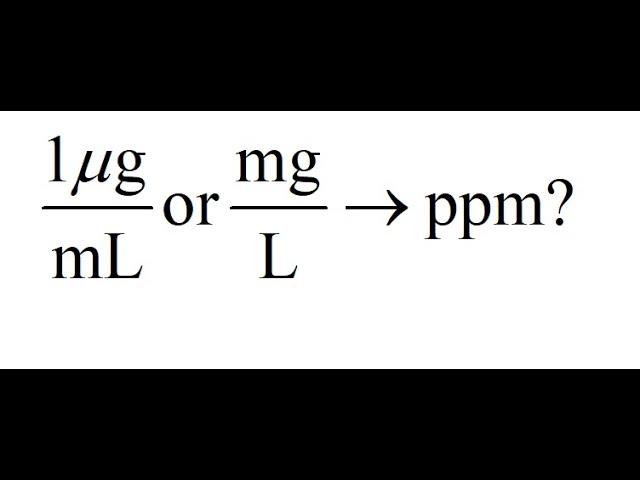10 ppm and 100 ppm Chloride Calibration Standard Sachets for HI9829 (10 x 25 mL each) - HI9829-12/13

SOLVED: a) Describe a preparation for a 100 mL, 220 ppm solution of NaCl. b) How would you prepare a 200 mL of 30 ppb solution of Iron c) Convert 25 ppm to M
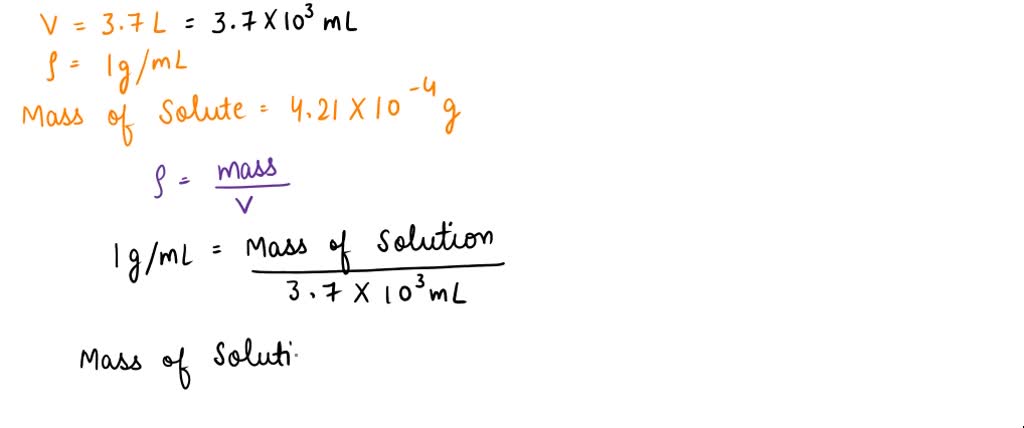
SOLVED: What is the concentration in ppm of a 3.7 L solution (d = 1.00 g/mL) containing 4.21x10^-4g of the pesticide DDT? (a) 0.114 ppm (b) 114 ppm (c) 11.4 ppm (d) 1.14 ppm
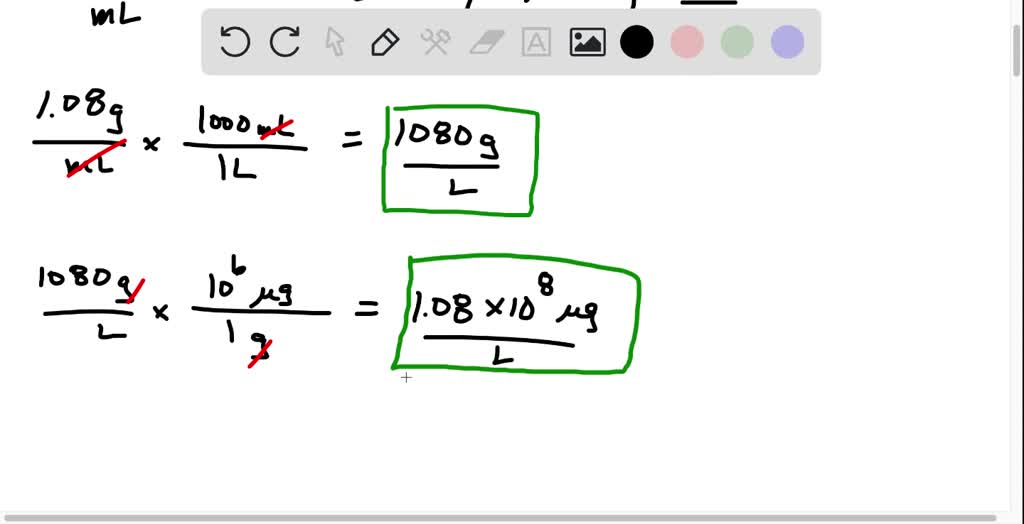
SOLVED: A dilute aqueous solution containing 1 ppm of solute has a density of 1.08 g/mL. Express the concentration of solute in g/L, μg/L, μg/mL, and mg/L.