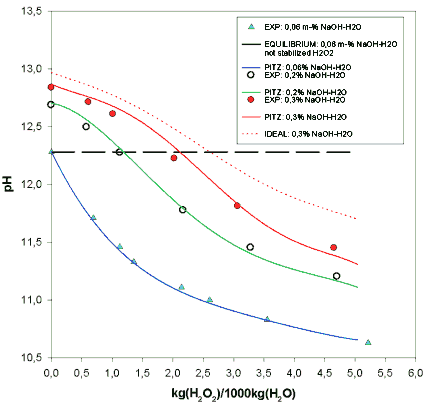
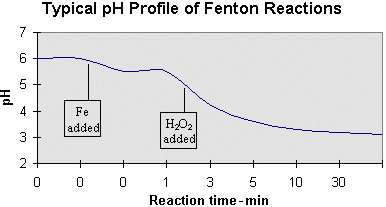
Sustainability | Free Full-Text | H2O2-Enhanced As(III) Removal from Natural Waters by Fe(III) Coagulation at Neutral pH Values and Comparison with the Conventional Fe(II)-H2O2 Fenton Process

Development of o-phthalic anhydride as a low-temperature activator in H2O2 bleaching system for cotton fabric | SpringerLink

Figure 10 from lectrochemical reduction of hydrogen peroxide on SIMFUEL ( UO 2 ) in acidic pH onditions | Semantic Scholar

SOLVED: Hydrogen peroxide, H2O2, dissociates in a manner similar to water producing H+ and HO2- ions. The pH of a 1M solution of hydrogen peroxide is 5.3 at 25C. Determine the value
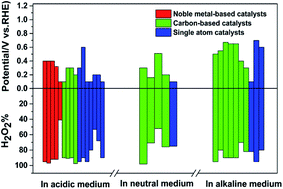
Electrochemical oxygen reduction for H2O2 production: catalysts, pH effects and mechanisms - Journal of Materials Chemistry A (RSC Publishing)

The life story of hydrogen peroxide II: a periodic pH and thermochemical drive for the RNA world | Journal of The Royal Society Interface

Temperature- and pH-Dependence of Hydrogen Peroxide Formation from Molecular Oxygen by Gold Nanoparticle-Loaded Titanium(IV) Oxide Photocatalyst | The Journal of Physical Chemistry C

Figure 12 from lectrochemical reduction of hydrogen peroxide on SIMFUEL ( UO 2 ) in acidic pH onditions | Semantic Scholar

Effects of pH and H2O2 on ammonia, nitrite, and nitrate transformations during UV254nm irradiation: Implications to nitrogen removal and analysis - ScienceDirect




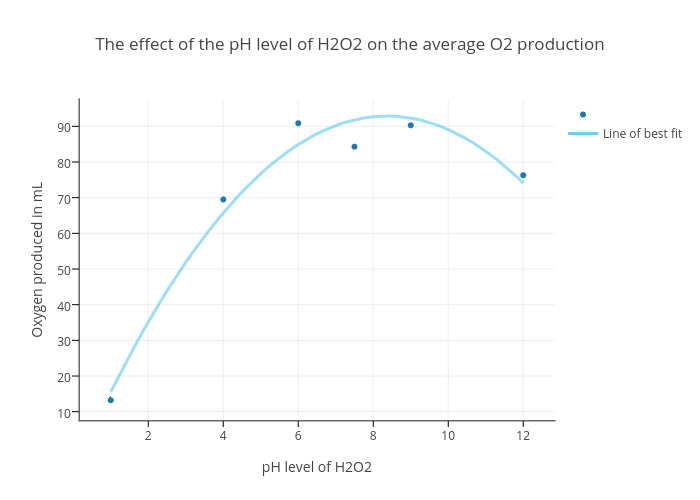
![Effect of pH Fig. 2: Effect of [H2O2] Fig. 3: Effect of [Fe 2+ ] | Download Scientific Diagram Effect of pH Fig. 2: Effect of [H2O2] Fig. 3: Effect of [Fe 2+ ] | Download Scientific Diagram](https://www.researchgate.net/publication/306039727/figure/fig23/AS:552945181618182@1508843812452/Effect-of-pH-Fig-2-Effect-of-H2O2-Fig-3-Effect-of-Fe-2.png)